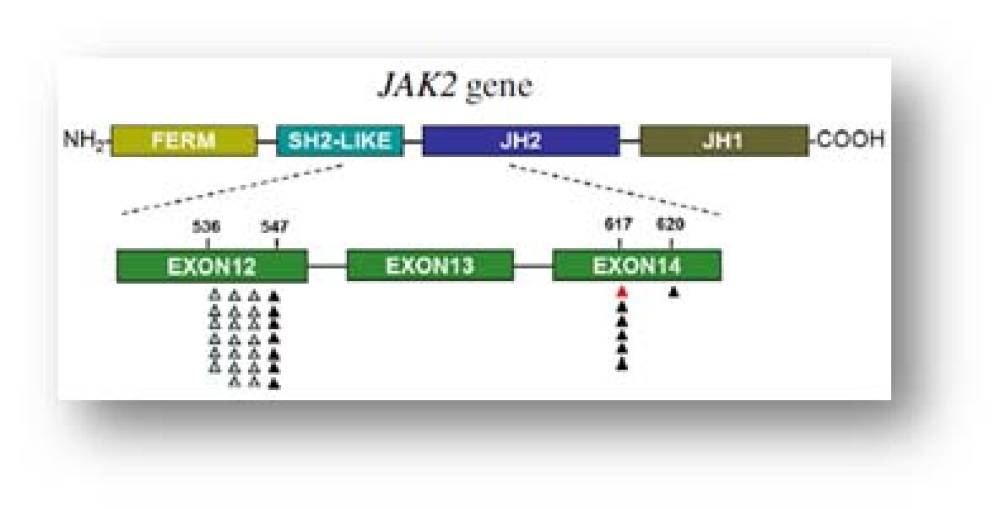
TRUPCR® JAK2 Kit (V2.0) is a real-time polymerase chain reaction (PCR) assay for qualitative detection of V617F mutation in JAK2 (Janus Kinase 2) gene against a background of wild-type genomic DNA.
This mutation is a guanine to thymidine transversion in position 1849 of the JAK2 gene, which leads to a valine to phenylalanine substitution in position 617 of the protein (V617F).
The TRUPCR ® JAK2 mutation kit is based on ARMS PCR. Taq DNA polymerase is extremely effective at distinguishing between a match and a mismatch at the 3’-end of a PCR primer. The kit is designed to selectively amplify mutant specific sequences in samples that contain a mixture of wild-type and mutated DNA.
Key Features
- The test allows highly sensitive site-specific detection of 0.5% mutant allele in a background of 99.5% wild-type allele.
- It is compatible with various Real Time PCR instruments.
- Easy-to-use, rapid, reliable, comprehensive and cost-effective tests.
- It is compatible with various Real Time PCR instruments.
- Complementary software analysis tool for easy detection and differentiation of mutants among wild-types.
Ordering Information:
| CAT. NO. | PRODUCT | CONTENTS |
|---|---|---|
| 3B1246 | TRUPCR® JAK2 KIT (Version 2.0) | 24 Reactions |
| 3B1247 | TRUPCR® JAK2 KIT (Version 2.0) | 48 Reactions |