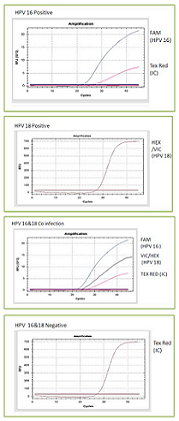
Test Overview:
TRUPCR® HPV 16 & 18 Detection Kit is a Real- Time PCR kit based assay for the qualitative detection of HPV 16 & 18 in clinical samples. This kit is based on amplification of the conserved region by primer and probes specific for HPV 16 & 18. In this kit there are three independent reactions running in arallel in two tube: the first tube detects HPV 16 (FAM channel), and internal control (IC) DNA (Texas Red) which allows excluding unreliable results and second tube detects HPV18 (HEX channel).
Advantages of TRUPCR® HPV 16 & 18 Detection Kit:
-
- Higher sensitivity and specificity
- Provided with internal control to exclude unreliable results
- Easy workflow & compatible with most of the real-time PCR equipment.
- All the reagents required for the test included in the kit.
About Human Papilloma Virus:
HPV is a small, non-enveloped, double-stranded DNA virus that replicates in the nucleus of quamous epithelial cells and induces hyper-proliferative lesions. HPV infections are among the most common sexually ransmitted infections. Most HPV infections have a benign clinical consequence and are cleared spontaneously. However, persistent HPV infection may result in progression to cervical cancer. More than one hundred different HPV genotypes have been identified, among which over forty infect mucosal and genital epithelia.
Genital HPV genotypes are generally classified into high risk (HR) and low risk (LR) groups based on their carcinogenic potential. Approximately 70% of invasive cervical cancer cases worldwide are caused by HPV 16 and HPV 18. Infection by HPV 16 or HPV 18 is associated with higher risk of disease progression compared to other HR HPV genotypes.
Ordering Information:
| CAT. NO. | PRODUCT | CONTENTS |
|---|---|---|
| 3B1276 | TRUPCR® HPV 16 & 18 Detection Kit | 48 Reactions |
| 3B1277 | TRUPCR® HPV 16 & 18 Detection Kit | 96 Reactions |
Publications:
- Narayanan, Geeta S., M. S. Ganesh, and Rishabh Kumar. "Comparison of treatment response in cervical carcinoma patients infected with human papillomavirus 16 and human papillomavirus 18 who are treated with chemoradiation." Journal of Cancer Research and Therapeutics 17.1 (2021): 204-210.
- Zheng, Ziwen, et al. "Prognostic value of HPV 16/18 genotyping and geminin mRNA quantification in low-grade cervical squamous intraepithelial lesion." Bioengineered 12.2 (2021): 11482-11489.
- Gupta, Shipra, et al. "Burden and associated genotype patterns of high-risk human papilloma virus infection and cervical cytology abnormalities among women in Central India." Infectious Diseases in Obstetrics and Gynecology 2022 (2022).
- Jwala, M., et al. "Prognostic Footprints of HPV Genotyping in Locally Advanced Carcinoma Cervix–A Single Institutional Study." International Journal of Radiation Oncology, Biology, Physics 114.3 (2022): e256-e257.
- John, Julie Hansa, et al. "Study to determine efficacy of urinary HPV 16 & HPV 18 detection in predicting premalignant and malignant lesions of uterine cervix." International Journal of Gynecology & Obstetrics 161.1 (2023): 79-85.
- Kulkarni, Sayali P., Shruti Paliwal, and Susmit Kosta. "Genotypic Diversity of Human Papillomavirus (HPV) Types and Its Prevalence With Cervical Cancer (CC) in Central India." Cureus 15.2 (2023).
- Khullar, Geeti, et al. "High-risk genital-mucosal human papilloma virus types 58 and 59 associated with solitary angiokeratoma on the elbow." Dermatology Practical & Conceptual 13.1 (2023).
- Patel, Sangram Singh, et al. "Comparison of Human Papillomavirus Genotype Detection in Paired Urine and Self-Collected Cervical Swabs: A Pilot Study." Asian Pacific Journal of Cancer Prevention 24.7 (2023): 2427-2430.
- JAYAPRAKASH, DR PARIKSHITH, and DR GEETA S. NARAYANAN. "Role of HPV DNA Testing and its influence on clinical outcomes in Cervical Cancer." Radiotherapy and Oncology. Vol. 133. ELSEVIER HOUSE, BROOKVALE PLAZA, EAST PARK SHANNON, CO, CLARE, 00000, IRELAND: ELSEVIER IRELAND LTD, 2019.