Respiratory Viruses in the Modern E...
05 Mar,2026
Globally, cervical cancer is the second most frequent malignancy in terms of both incidence and mortality in women of reproductive age 15 to 44 years, with a particularly high burden in many countries. Notably, India accounts for 9.0% of cervical cancer incidences and 8.7% of deaths.
The natural history of cervical cancer is well understood, including a HPV infection, progression to pre-cancer, and invasion to cancer. Other risk factors, including smoking, HIV infection, early sexual debut, oral hormonal contraceptives, and multiple sexual partners, collectively contributed a large number of cervical cancers. All these facts make cervical cancer a largely preventable disease. The main cause of cervical cancer is persistent chronic Human Papillomavirus (HPV) infection, and a combination of HPV types 16 and 18 caused 71 % of cervical cancers worldwide. HPV is the most common sexually transmitted infection in the world and is classified as a carcinogenic infectious agent by the International Agency for Research on Cancer. However, only some HPV strains are oncogenic. Approximately 99.7% of cervical cancers, 90% of anal, 65% of vaginal, 50% of vulvar, and 45-90% of oropharyngeal cancers are caused by the HPV infection.
More than 100 different HPV genotypes have been identified, and more than 40 types are known to be sexually transmitted and to affect anogenital epithelium. Anogenital infections are associated with about 40 HPV genotypes, which are classified into high, low, and intermediate risk types according to their carcinogenic potential. Due to their involvement in high-grade squamous intraepithelial infection (HSIL) or cervical cancer, HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, 73, and 82 are classified as high-risk or oncogenic types, while HPV 6, 11, 40, 42, 43, 44, 54, 61, 70, 72, and 81 are referred to as low grade types and are associated with benign warts and HPV 23, 53, and 66 as intermediate risk types.
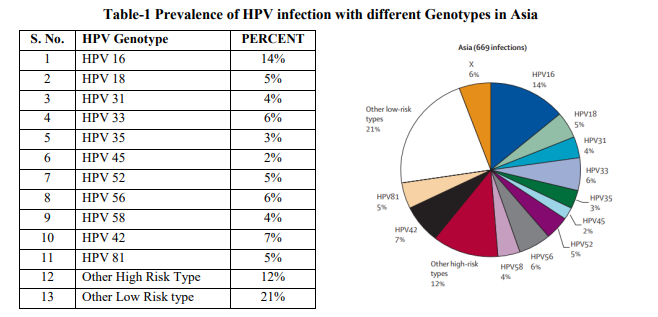
Table 1 shows the prevalence of HPV infection with different Genotype in Asia region. HPV16 was twice as frequent as any other high-risk type. The next most common high-risk types were HPV33 and HPV56 in Asia.
“World Health Organisation (WHO) Guidelines for Screening and Treatment of Cervical Pre-cancer Lesions for Cervical Cancer Prevention” recommends the use of DNA-based HPV testing as a first choice screening method because these are more efficient than cytology-based HPV tests, especially for low and middle-income countries; as they are less prone to quality problems. Therefore, identifying at risk individuals through HPV testing enables earlier intervention, appropriate monitoring, and supports vaccination strategies.
WHO proposes this global strategy to eliminate cervical cancer:

Compared with cervical screening methods identifying cytological abnormalities, molecular tests that specifically detect the presence of HR HPV DNA in cervical cells can potentially increase sensitivity and cost-effectiveness of cervical cancer screening. Real-time Polymerase Chain Reaction (RT-PCR) is the gold standard for HPV diagnosis. “TRUPCR® HPV High Risk Genotyping Plus Kit” is based on the Real-Time PCR system for the qualitative detection and genotyping of 14 High Risk HPV DNA and 2 Low Risk HPV DNA in clinical samples such as vaginal swabs, urine, and formalin-fixed, paraffin-embedded (FFPE) tissue. This TRUPCR® HPV HR Kit is CE-IVD approved.
TRUPCR® HPV High Risk Genotyping Plus Kit is a Real-Time Amplification test for the qualitative detection of Human Papilloma Virus (HPV) DNA in clinical samples. This kit is based on amplification of E6/E7 region by primer and probes specific for HR HPV 14 Genotypes (16/18/31/33/35/39/45/51/52/56/58/59/66). For LR HPV 6 & 11, the primers and probe are based on LCR and E4 protein, respectively. The kit includes Master Mix and primer-probe mix for the specific amplification of E6/E7 region of the HPV HR (High Risk) genome, as well as a positive and a negative control to confirm the integrity of the kit reagents. In this kit there are six independent reactions running parallel in 6 tubes. Details are as follows.
| Tube | HPV Genotyping | Detector channel | Reporter | Quencher |
| 1 | HPV 59 | Green | FAM | None |
| HPV 18 | Yellow | HEX/VIC | None | |
| HPV 56 | Orange | TEXAS RED/ROX | None | |
| 2 | HPV 33 | Green | FAM | None |
| HPV 31 | Yellow | HEX/VIC | None | |
| HPV 35 | Orange | TEXAS RED/ROX | None | |
| 3 | HPV 58 | Green | FAM | None |
| HPV 45 | Yellow | HEX/VIC | None | |
| HPV 68 | Orange | TEXAS RED/ROX | None | |
| 4 | HPV 52 | Green | FAM | None |
| HPV 39 | Yellow | HEX/VIC | None | |
| HPV 51 | Orange | TEXAS RED/ROX | None | |
| 5 | HPV 16 | Green | FAM | None |
| HPV 66 | Yellow | HEX/VIC | None | |
| Endogenous Internal Control | Orange | TEXAS RED/ROX | None | |
| 6 | HPV 6 | Green | FAM | None |
| HPV 11 | Yellow | HEX/VIC | None |
In 2022, Gupta et al., conducted a study on 295 women in central India; TRUPCR® HPV HR with 16/18 Genotyping Kit was used and found that HPV 16 (n=29; 60.4%) was the most common genotype, HPV 18 was detected only in 6.25%, whereas other high-risk HPV genotypes were found to be 12.5%, mixed infections of HPV was detected in 20.8%.
Further, A study was conducted in a tertiary healthcare institution in India and included 180 women aged 35– 65 years. Additionally, Kulkarni et al., performed a study on 736 women in different age group and reported that HPV-16, 18, 31, and 45 each had a prevalence of 29.6%, 11.1%, 12.9%, and 9.2%, respectively, while the overall prevalence of HR-HPV was present in populations at 7.3% in individuals and 37.0% in combinations. HR-HPV infection and prevalence were provocatively more (79.6%) in the Visual inspection with acetic acid (VIA) positivity rate with cervical cancer.
Another study, which is conducted by the Yadav et al., using TRUPCR® HPV High Risk Genotyping Plus Kit the in a total of 185 samples were analysed by Real-Time PCR, out of which 43 samples were positive for HPV. In which HPV16 accounts for 62.79% of the total prevalence followed by HPV58 (9.3%), HPV31, HPV33, and HPV39 each having prevalence of 6.97%.
05 Mar,2026
27 Jan,2026
27 Jan,2026
24 Jan,2026
24 Jan,2026
3B BlackBio Biotech India Limited is now 3B BlackBio Dx Limited as a result of amalgamation with it's parent company Kilpest India Limited. 3B BlackBio Dx is a leading Indian company in the field of PCR based Molecular Diagnostic Kits. We offer technical support and training on all our products and are committed to increasing the efficiency of laboratory testing and enhancing patient care.
The Company is ISO 13485:2016 certified, GMP compliant biotech R&D organization.
General enquires: info@3bblackbio.com
Corporate Enquiry:
+91 9691680693,+91 9810562700
Orders: info@3bblackbio.com
Tel: 0755 - 4076518, 4077847