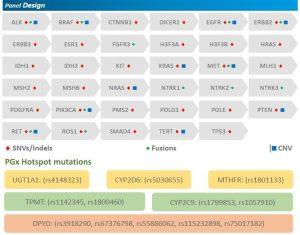
The Solid Tumor Panel is designed to detect SNVs, Indels CNVs and RNA fusions in 35 marker genes and hotspots in 6 pharmocogenomics genes associated with solid tumors such as lung, gastro intestinal/colorectal, breast, liver and ovarian tumors.
For use on Illumina and MGI platforms only.
Specifications
- Starting Material: 50-100 ng of DNA, 300-600 ng RNA
- Sample source: FFPE, Fresh Frozen Tissue
- Target region for DNA Panel: 117 kb
- Target region for RNA Panel: 74 kb
- Library preparation time: 1.5 days
- Bioinformatics Analysis: 1.5 hrs (from FASTQ to report)
Key Features:
- Carefully curated panel of 35 genes with Clinical Actionability ( mutations especially relevant to lung and colorectal cancers)
- Designed as per NCCN and ESMO Guidelines
- Detects SNV/INDELS, CNV and RNA Fusions
- Uses Hybrid Capture technology to ensure full coverage of all exonic regions of each gene and allows for the discovery of novel mutations/alterations
- Includes hotspot for six clinically relevant pharmacogenomics genes DPYD, UGT 1 A 1, CYP 2 D 6 MTHFR, TPMT and CYP 2 C 9
- LoD as low as 3 5 % (calculated for SNV using reference material at 500 X)
Ordering Information:
| CAT. NO. | PRODUCT | CONTENTS |
|---|---|---|
| 3BNGS001-16 | TruNGS® Solid Tumor Panel Kit | 16 Reactions |
| 3BNGS001-32 | TruNGS® Solid Tumor Panel Kit | 32 Reactions |
| 3BNGS001-48 | TruNGS® Solid Tumor Panel Kit | 48 Reactions |