When Your Genes Become the Enemy: U...
18 May,2026
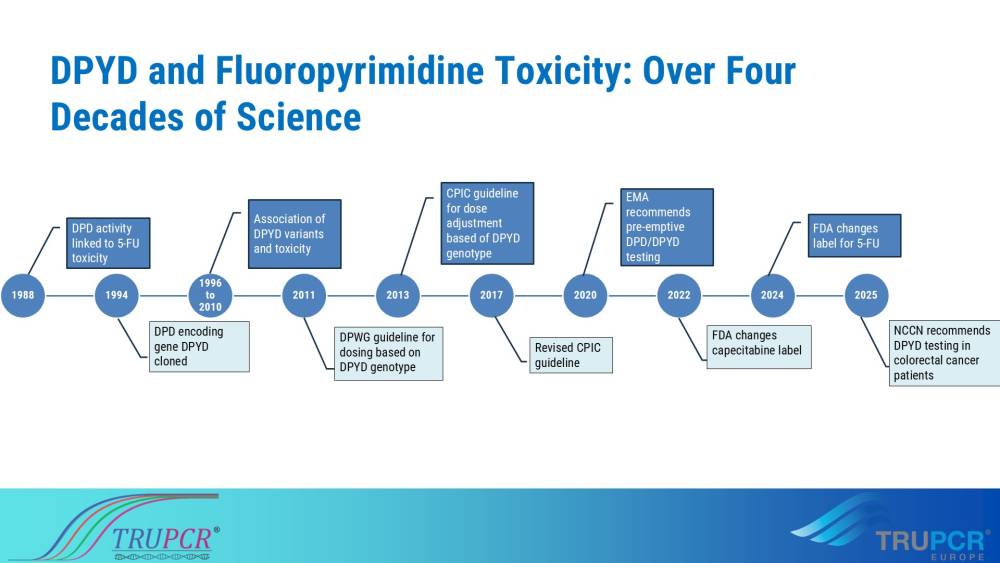
India reported 1,461,427 new cancer case in 2022 which is suggesting that 1 in every 10 people are at risk to develop cancer by the age of 74. This number is projected to increase by 12% yearly making India one of worse hit country. Chemotherapy is mainstay for several cancers, however, not every chemotherapy agent works equally well for all the patients receiving them. Some people may experience adverse drug reaction and may even succumb to it. This is due to variation in genes involved in detoxifying these drugs. One such well- studied and well-stablished gene drug interaction is Fluoropyrimidine and DPYD. Fluoropyrimidines such as 5-fluorouracil (5-FU) and its prodrug capecitabine are widely used as adjuvant chemotherapy to treat many different neoplasms such as breast cancer, head and neck squamous carcinoma and colorectal cancers. Discovered in 1950s by Heidelberger and co-workers, 5-FU is still the backbone of many chemotherapies, especially in colorectal cancers. A global market size of 2.45 billion in 2023 and projected to reach USD 4.34 billion by 2030 further testifies the utility of 5-FU in cancer treatment and management. However, not everything is bright and shiny with this relatively cheap and effective drug, some individuals may experience adverse drug reaction and fewer some may even succumb to death.
Over 50% of 5-FU related drug toxicity could be attributed to the genetic variations in the gene encoding Dihydropyrimidine dehydrogenase (DPD) enzyme, DPYD. DPD enzyme is responsible for metabolism of 5-FU. Certain genetic variations in DPYD gene can reduce or completely inactivate the enzyme affecting the metabolism of the drug. Several studies for the last 40 years have identified and stablished links between DPYD genetic variations and fluoropyrimidine chemotherapy toxicity in patients. These variations are found in 3-5% of individuals globally. Realizing the importance DPYD genetic variations, Dutch Pharmacogenetics Working Group (DPWG) recommended pre-emptive testing for DPD deficiency and adjusting the dose of fluoropyrimidine to avoid toxicity as early as 2011. As the evidences keep mounting in favour of pre-emptive DPYD testing to avoid/reduce chemotherapy toxicity, several other national and international bodies such as Clinical Pharmacogenetics Implementation Consortium (CPIC®), European Medicine Agency and others recommend dose adjustment based on DPYD genotype of the patient. While several studies and publicly available genomic data from India suggest a similar or higher prevalence of DPYD deficiency variants in Indian population, use of pre-emptive DPYD genotyping before starting 5-FU chemotherapy is abysmal at best in the country. Several factors, such as the lack of specific guidelines, no insurance coverage and poor awareness of the importance of DPYD testing in avoiding drug toxicity contribute to this poor acceptance of a well-established standard of care practice. It is high time we prioritize patient safety and adapt tools of personalized medicine to provide best possible care to people who need it the most, that’s everyone undergoing fluoropyrimidine chemotherapy. Simply, because every life is worth fighting for.
18 May,2026
24 Apr,2026
16 Apr,2026
07 Apr,2026
07 Apr,2026
3B BlackBio Biotech India Limited is now 3B BlackBio Dx Limited as a result of amalgamation with it's parent company Kilpest India Limited. 3B BlackBio Dx is a leading Indian company in the field of PCR based Molecular Diagnostic Kits. We offer technical support and training on all our products and are committed to increasing the efficiency of laboratory testing and enhancing patient care.
The Company is ISO 13485:2016 certified, GMP compliant biotech R&D organization.
General enquires: info@3bblackbio.com
Corporate Enquiry:
+91 9691680693,+91 9810562700
Orders: info@3bblackbio.com
Tel: 0755 - 4076518, 4077847